School of Life Science and Technology Associate Professor Tiffany Horng published a review article entitled Metabolism as a guiding force for immunity in Nature Cell Biology in January 2019, in collaboration with Mayo Clinic Assistant Professor Hu Zeng to publish a review article. This article reviews recent studies that have elucidated how the metabolism of immune cells plays an important role in immune activation. Indeed, the relationship between immunity and metabolism has become a hot topic of biology research in recent years.
Immune cells are divided into myeloid and lymphoid lineage, and the article introduces the metabolism of myeloid cells represented by macrophages and lymphocytes represented by T cells. Assistant Professor Horng was responsible for reviewing the activation and metabolism of macrophages.
Macrophages are innate immune cells that act as first-in-command in defense against invading pathogens. Lipopolysaccharide (LPS) on the surface of Gram-negative bacteria can stimulate macrophages to the M1 state, leading to production of inflammatory cytokines and antimicrobial peptides critical for bacterial restriction, in a process called classical activation. IL-4 cytokine secreted by helper T cells can activate macrophages to the M2 state, leading to upregulation of tissue repair, in a process called alternative activation. The review describes how macrophage metabolism plays a crucial role in guiding and regulating such macrophage activation. In classical activation, glucose utilization and aerobic glycolysis are upregulated but oxidative metabolism is turned off, while oxidative metabolism is increased in alternative activation. Genetic and pharmacological manipulations that disrupt these metabolic changes impair macrophage activation, indicating a key role for metabolism in guiding and regulating macrophage biology.
The review also discusses how the metabolism of macrophages differs in distinct tissues, and can influence the activities of surrounding cells like T cells. Overall, the review highlights how recent studies in immunometabolism have revealed that cellular metabolism is not only a basic process for maintaining cell life, but also a highly adaptive guiding force in cell fate determination.
In November last year, Tiffany Horng joined the School of Life Science and Technology in November 2018, having previously served as independent investigator at Harvard T.H. Chan School of Public Health since 2008. Her lab’s research direction uses macrophages as model system to systematically explore the teleological rationale and underpinning mechanisms of metabolic support of macrophage biology.
Article link: https://www.nature.com/articles/s41556-018-0217-x
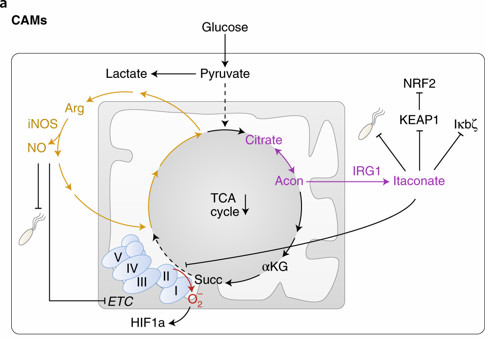
Metabolism of macrophages in the classical activation
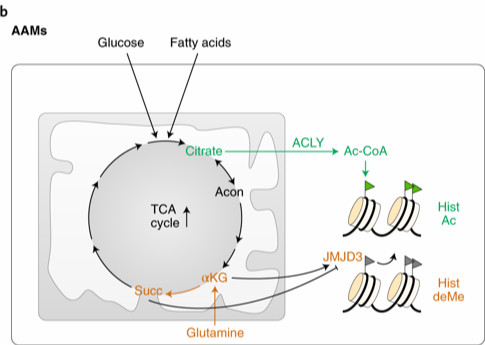
Metabolism of macrophages in the alternative activation


